Welcome to our company !
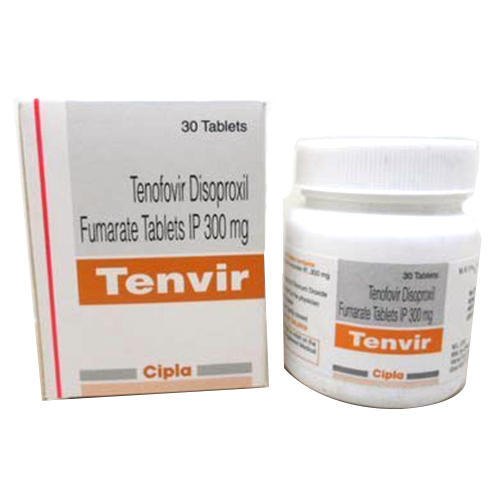
Tenvir 300mg Tablet
Product Details:
- Medicine Type Allopathic
- Indication HIV, Chronic Hepatitis B Virus infection
- Grade Pharmaceutical Grade
- Storage Store below 30C, protect from moisture
- Expiration Date 36 months from manufacturing date
- Dosage Form Tablet
- Pacakaging (Quantity Per Box) 30 tablets
- Click to View more
X
Tenvir 300mg Tablet Price And Quantity
- 100 Tablet
Tenvir 300mg Tablet Product Specifications
- Allopathic
- HIV, Chronic Hepatitis B Virus infection
- Pharmaceutical Grade
- 36 months from manufacturing date
- Store below 30C, protect from moisture
- Tablet
- 30 tablets
- Film-coated tablet
- Not less than 98% and not more than 102% of Tenofovir Disoproxil Fumarate
- 147127-20-6
- Tenofovir Disoproxil Fumarate 300mg
- India
- C19H30N5O10PC4H4O4
- Treatment of HIV-1 infection and chronic hepatitis B
- 300 mg
- Prescription
- White
- Adults and adolescents
- Cipla Ltd.
- WHO-GMP, FDA approved
- Nausea, vomiting, diarrhea, headache, dizziness
- 3 years
- Equivalent to Tenofovir 245 mg
- Bottle
- HDPE bottle
- Oral
Product Description
Experience splendiferous health benefits with Tenvir 300mg Tablet-currently on a limited time special sale! Manufactured by Cipla Ltd. and packaged in immaculate HDPE bottles, these enviable white, film-coated tablets offer prescription-grade efficacy for the treatment of HIV-1 infection and chronic hepatitis B. Each tablet provides pharmaceutical-grade Tenofovir Disoproxil Fumarate (300mg), ensuring an assay of not less than 98% and not more than 102%. With a shelf life of 3 years, WHO-GMP and FDA approval, Tenvir delivers consistent quality and safety.
Impressive Application and Advantages of Tenvir 300mg Tablet
Tenvir 300mg Tablet is prescribed for adults and adolescents in the management of HIV-1 infection and chronic hepatitis B. With a pharmaceutical-grade composition and WHO-GMP, FDA approvals, it stands out for its enviable purity, efficacy, and reliability. Its specific usage allows targeted antiviral therapy, while competitive advantages include an immaculate safety profile, long shelf life, and film-coated, easy-to-administer dosage form-making Tenvir a preferred choice among healthcare professionals.
Efficient Delivery, Export Market Reach, and Sample Availability
Tenvir 300mg Tablet is always stock ready and dispatched promptly to national and international markets. Our packing and dispatch process ensures the product reaches its destination in optimal condition, preserving quality and safety. Main export markets include Asia, Africa, and Latin America, reflecting our global presence. Samples are available upon request, ensuring confidence in product selection. Quick delivery and responsive service make Tenvir an excellent option for distributors, exporters, and suppliers.
Impressive Application and Advantages of Tenvir 300mg Tablet
Tenvir 300mg Tablet is prescribed for adults and adolescents in the management of HIV-1 infection and chronic hepatitis B. With a pharmaceutical-grade composition and WHO-GMP, FDA approvals, it stands out for its enviable purity, efficacy, and reliability. Its specific usage allows targeted antiviral therapy, while competitive advantages include an immaculate safety profile, long shelf life, and film-coated, easy-to-administer dosage form-making Tenvir a preferred choice among healthcare professionals.
Efficient Delivery, Export Market Reach, and Sample Availability
Tenvir 300mg Tablet is always stock ready and dispatched promptly to national and international markets. Our packing and dispatch process ensures the product reaches its destination in optimal condition, preserving quality and safety. Main export markets include Asia, Africa, and Latin America, reflecting our global presence. Samples are available upon request, ensuring confidence in product selection. Quick delivery and responsive service make Tenvir an excellent option for distributors, exporters, and suppliers.
FAQ's of Tenvir 300mg Tablet:
Q: How should Tenvir 300mg Tablet be administered?
A: Tenvir 300mg Tablet is taken orally, usually once daily, as prescribed by a healthcare professional. Swallow the tablet with water, and do not crush or break it.Q: What is the main benefit of using Tenvir 300mg Tablet?
A: Tenvir offers effective treatment of HIV-1 infection and chronic hepatitis B due to its high purity active ingredient and consistent dosage, helping improve patient health outcomes.Q: What side effects may occur with Tenvir 300mg Tablet?
A: Common side effects include nausea, vomiting, diarrhea, headache, and dizziness. If any severe reactions occur, seek immediate medical advice.Q: Where is Tenvir 300mg Tablet manufactured and who markets it?
A: Tenvir 300mg Tablet is manufactured in India and is marketed by Cipla Ltd., supported by international standards such as WHO-GMP and FDA approvals.Q: When does Tenvir 300mg Tablet expire and how should it be stored?
A: The shelf life is 36 months from the date of manufacturing. Store the tablets below 30C, protected from moisture, in their original HDPE bottle.Q: What is the process for obtaining a sample of Tenvir 300mg Tablet?
A: Samples are available for evaluation. Distributors, exporters, and suppliers can request samples by contacting the marketing or export team at Cipla Ltd.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese

 Send Inquiry
Send Inquiry Send SMS
Send SMS
